Tenvir EM is a combination medicine containing Tenofovir disoproxil fumarate and emtricitabine as the active ingredients. It is used as a part of a regimen of antiretroviral medications to treat HIV-1 in adults. The medication is taken along with safer sex practices to reduce the risk of sexually acquired HIV-1 in adults aged over 18 years at high risk.
The combination drug comes in tablet form, and these tablets are used in combination with other medications that have a different working mechanism to treat HIV patients. Such patients have either already received treatment with other HIV medication or are undergoing first-line treatment. Both antiretroviral agents are effective against RNA viruses such as Human Immunodeficiency Virus (HIV).
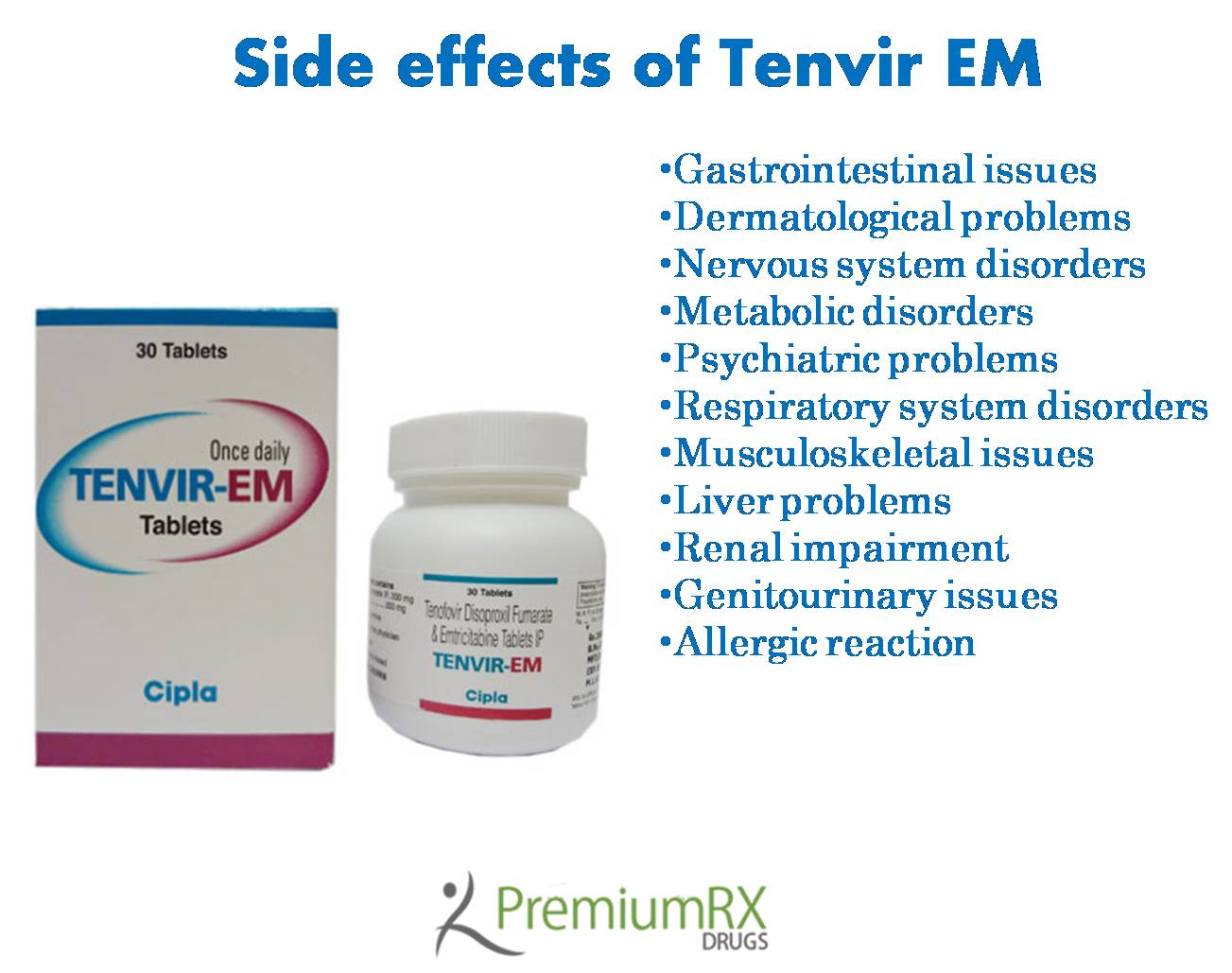
Side effects associated with Tenvir EM
Along with the benefits, like other pharmaceutical agents, Tenvir EM also comes with side effects. The most commonly reported side effects reported with the active ingredient Tenofovir disoproxil fumarate in HIV-1 infected patients include diarrhea, pain, asthenia, nausea, rash, and headache. Clinical trials were conducted to check the range of side effects in people taking this combination medicine. During the controlled clinical trial, the most common side effects of this antiretroviral agent include mild to moderate gastrointestinal issues such as vomiting, diarrhea, nausea, and flatulence.
In patients with chronic hepatitis B virus (HBV) infection and hepatic insufficiency, side effects included diarrhea, fatigue, pain, skin rash, back pain, headache, nausea, dizziness, abdominal pain, and nasopharyngitis were reported with Tenofovir disoproxil fumarate.
Gastrointestinal issues- The gastrointestinal events associated with the combination drug include nausea, vomiting, and abdominal pain, most commonly occurring in patients who are suffering from HBV and decompensated liver disease. Pancreatitis has also been reported during postmarketing experience.
Common side effects are diarrhea, vomiting, nausea, and abdominal pain. Pancreatitis and elevated serum lipase are some uncommon side effects of the medicine.
Dermatological problems-
Very common ones include rash events such as pruritus, exfoliative rash, macular rash, vesicular rash, urticaria, and maculopapular rash. The commonly reported dermatological issue was sweating.
Nervous system disorders-
Very common side effects associated with Tenvir EM include dizziness and headache, while peripheral neuropathy (a medical condition that affects the nerves, impairing sensation movement and/or organ function) is also common. Dizziness was also reported in patients with chronic HBV and decompensated hepatic impairment.
Metabolic disorders-
- Lactic acidosis and severe hepatomegaly (enlarged liver) with steatosis, also called fatty liver, have been reported with the use of this medication.
- Very common side effects associated with using Tavin EM 300/200mg include hypophosphatemia, elevated fasting cholesterol, and elevated triglycerides.
- The common ones are anorexia, elevated alkaline, and increased fasting triglycerides. In contrast, the rare side effects include lactic acidosis and hypokalemia (potassium deficiency in the blood).
Psychiatric problems-
Insomnia was reported in patients with chronic HBV and decompensated hepatic impairment. Depression, insomnia, abnormal dreams, and anxiety were commonly reported side effects of the antiretroviral drug.
Respiratory system disorders-
- Common- infection of the upper respiratory tract, pharyngolaryngeal pain, pneumonia, nasopharyngitis (commonly called a cold)
- Uncommon- nasal congestion
Musculoskeletal issues–
- Common: back pain, arthralgia (joint pain)
- Uncommon ones: muscular weakness
- Rare one- myopathy (a disorder of muscle tissue)
Liver problems-
Severe acute exacerbations of hepatitis have been reported in patients with hepatitis B after stopping the HIV treatment with Tenvir EM.
Renal impairment-
Common: Increased creatinine levels
The rare ones include acute renal failure, renal failure, nephrogenic diabetes insipidus, and acute tubular necrosis.
Genitourinary issues-
- Common- glycosuria (excretion of glucose into the urine), hematuria (Presence of blood in urine)
- Uncommon- decreased urine volume
Allergic reactions such as angioedema were also reported in the clinical trials.
Latest posts by Marie (see all)
- Acid Reducer Medications: Types, Benefits, and How They Work - June 10, 2026
- A Complete List of Asthma Inhaler Names for Asthma Management - June 9, 2026
- Top Natural Alternatives to Tadalafil: What Actually Works? - June 5, 2026